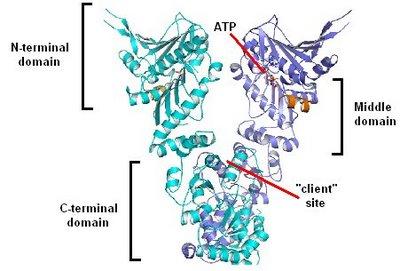
Moving away from the specifics of Hsp90 for a while, this post shall focus on the general principle of chaperones in evolution. What supports such an hypothesis:
To begin lets look at the problem of rapid evolution. Darwinian evolution is based on a constant rate of random mutations in the genome of any evolving organism. This implies that mutations happens constantly, by chance, regardless of the external environment. Adaptations consequently arise by chance. Evolution of new traits one may think, is therefore slow and gradual.

Gradual evolution (reproduced with permission from Dr. Dennis O’Neil)
Since such a view, does not fit with the bursts of evolution observed in fossil material, alternative explanations have been put forward (see more below). However, even when using a constant random rate of mutations one would expect “bursts” or rapid transitions. This is elegantly illustrated in this simulation of an evolving clockwork. Since a beneficial mutation can have a profound impact on fitness, then there should be no surprise that the transition between av less fit form and a more fit one, happens quickly. Thus even with a constant mutation rate one would probably not see a slow, gradual evolution of species, – basic math skills on exponential growth should make this clear. Why this notion of gradual evolution is prevailing I cannot understand.
In addition there are those that believe that the mutation rate may not be constant. Thus, with an increased mutation rate and rapid transitions one can start to explain the observed bursts of evolution.
Further explaining bursts of evolution we have the theory of ‘punctuated equilibria’, associated with Niles Eldredge and Stephen Jay Gould, which states that organisms go through short periods of rapid evolution from time to time, against a background of relative stasis (see picture below, and this genomicron post as a starting point for more on punctuated equilibrium ).
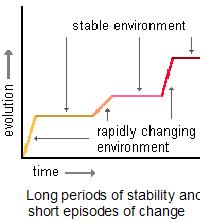
Punctuated equilibrium (reproduced with permission from Dr. Dennis O’Neil, for his tutorials on evolution go here and here).
This has further led to the theory of hopeful monsters. These theories account for non-linear rapid evolution within the boundaries of Darwinian principles, but they have been heavily criticed. One of the main criticisms of these theories, as far as I can understand, is the improbability of a single mutation to give rise to radical morphological changes, and further that this change, if it happens, is most likely deleterious, and if it against all odds is beneficial, its even more improbable that this individual is able to produce offspring with the same trait(s).
So we are still left with some problems: External environment changes can happen really quickly. Is random mutation events, occurring at a slow rate (even if it’s sped up in larger populations or even if monster are hopeful in times of stress), sufficient to explain the effectiveness of adaptations seen in nature ? Does an organism rely on (slow) random mutations to evolve a trait to help the species adapt to the new environment, or are there additional mechanisms in place to speed up this mutation rate and perhaps guide mutation events towards selected genes that allows rapid changes in phenotypes ?
Enter heat shock proteins…….
The hypothesis is the following: If there is a way to mask (deleterious) changes in proteins under normal conditions, one may accumulate such changes without exposing them.
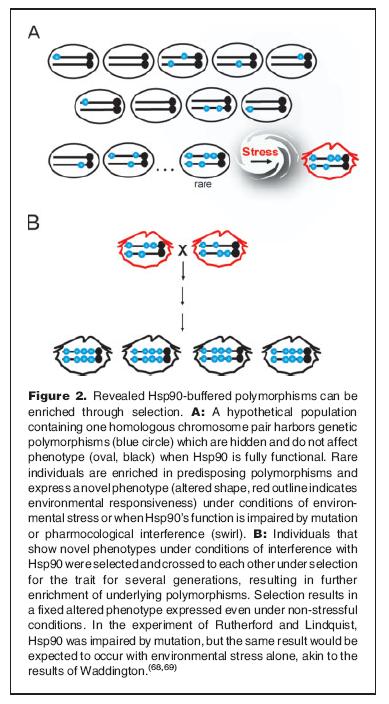
Illustration from Sangster TA et al. (more on Waddington will follow in the last post).
Thus, with Hsp90 acting as a buffer: one could potentially get a lot of hopeful monsters, under times of stress, as these traits were exposed. This would drastically increase the chances of a beneficial change to occur at the right time. And since the chance of mating with other monsters with similar traits (there are more than one monster, in fact very many), the chance of keeping the trait(s) in subsequent generations is also increased.
Now missing……..evidence, which will follow in the next post.