Hsp90 and it’s possible role in evolution (as a capacitor for rapid change), I have covered extensively in the 5 post series for JustScience week 08 (Revolution Evolution, Presenting….Hsp90, How can chaperones act in evolution, Evidence for Hsp90 involvement in rapid evolution of new traits and Hsp90 to end controversies in evolution theory). Recently I found this thorough review on the subject from which I would like to share the essentials (review written by Roberta L Millstein at University of California, Davis):
Recent work on the heat-shock protein Hsp90 by Rutherford and Lindquist …. has been included among the pieces of evidence taken to show the essential role of developmental processes in evolution;
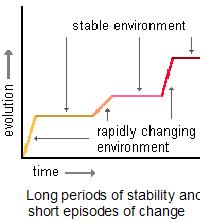
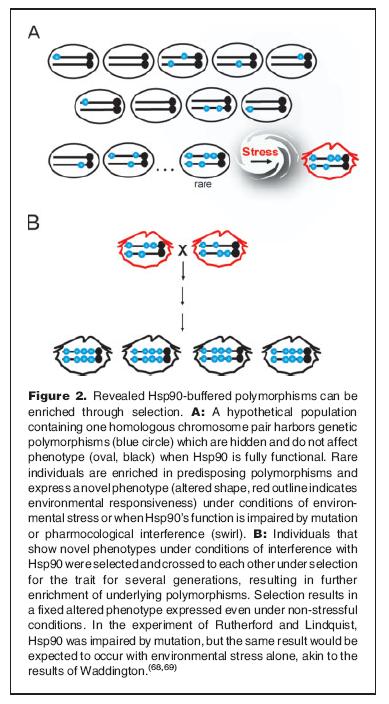
To recap, the theory is that heat shock proteins can hide genetic variation until a stressful environment exposes them to allow rapid change (evolution) of morphology and subsequently, traits.
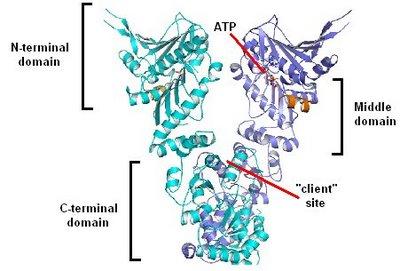
Hsp90 acts as a buffer against phenotypic variation, allowing genotypic variation to build. When the buffering capacity of Hsp90 is altered (e.g., in nature, by mutation or environmental stress), the genetic variation is “revealed,” manifesting itself as phenotypic variation.
The theory is backed up by genetic experiments on Drosophila and Arabidopsis. Results from this research on Hsp90 lends support to channeling and “hopeful monster” theory and as such, follows the more controversial line of evolution-thinking. The review sums up many of the controversial sides of conclusions from the Hsp90 research:
This phenomenon raises questions about the genetic variation before and after what I will call a “revelation event”: Is it neutral, nearly neutral, or non-neutral (i.e., strongly deleterious or strongly advantageous)? Moreover, what kinds of evolutionary processes do we take to be at work?
My goal with the previous posts on Hsp90 was to show that the data lends sufficient support to alter and revise how we think about evolution. It seems this is the goal of the review as well.
The primary goal of this paper is to illuminate the alternative scenarios and the processes operating in each. At the end, I raise the possibility of a synthesis between evo-devo and nearly neutral evolution.
Evolution I strongly believe, is not entirely and exclusively driven by a random (and slow) constant mutation rate, but rather controlled by a number of additional mechanisms to ensure that an organism can evolve rapidly. To me, this is not controversial at all, – it does not overturn any Darwinian principles, but serves as an extention to explain the speed of evolution that has sometimes baffled us. Nevertheless, conclusions drawn from Hsp90 research remains controversial to many evolutionists, and Millstein sums it up with:
I find it somewhat ironic that people who are otherwise unorthodox in their thinking with respect to evolution are so orthodox when it comes to adaptationism. After all, as the late Gould argued, nonadaptive approaches were left out of the evolutionary synthesis (Gould 1983) just as developmental processes were (Gould 2002).
Which to me, a molecular biologist gone amateur evolutionist, is a good ending note. Reviews like this, one can only hope, will lend credibility to alternative thoughts on mechanisms of Darwinian evolution. Which is surely needed to fully understand the beauty and complexity of the molecular mechanisms that shapes our world.
Note: the review I have linked to is open access, but apparantly only a draft, the final version is available here, but isn’t open access (shame on you Biological Theory and MIT press Journals).