Chapter I gave an introduction into the role of Heat Shock Protein 90 (Hsp90) in evolution. The main point was its ability to mask/hide transforming mutations until needed in stressful times. This chapter describes its physical and biological properties.
Firstly, what the protein looks like. A really good introduction into Hsp90’s structure is found at the Sandwalk blog. Here’s a picture I stole (with permission) from his blog post on Hsp90:
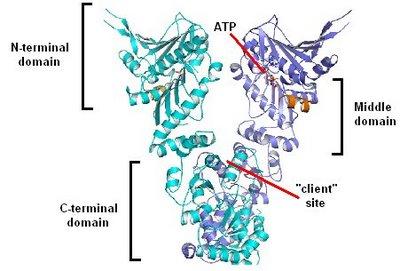
Picture from: Dollins, E.D., Warren, J.J., Immormino, R.M. and Gewirth, D.T. (2007) Structures of GRP94-Nucleotide Complexes Reveal Mechanistic Differences between the hsp90 Chaperones. Molec. Cell 28:41-56.
From Sandwalk: The complete protein is a dimer of two identical subunits. Each monomer has three distinct domains; an N-terminal domain (N); a middle domain (M); and a C-terminal domain (C). The ATP hydrolysis site sits at the interface between the N and M domains. The C domains interact to form the dimer. The presumed site of binding for misfolded proteins (“client” site”) is in the V-shaped pocket formed when the C domains come together. The mechanism of action of Hsp90 proteins is not known although it presumably involves a conformational change induced by ATP hydrolysis.
Secondly, it’s mode of action. Hsp90 works in concert with many other proteins to form protein-complexes ultimately activating the target protein. The molecular mechanism is as mentioned, still unresolved despite the presence of crystal structures. However, the following illustrations are taken from The Jackson Laboratory at University of Cambridge UK, to depict the overall events:
 |
|
Model of the activation of a client protein by chaperone machines.
The chaperone Hsp70 system targets the client protein, in this case the steroid receptor, to the Hsp90 chaperone via the organising protein HOP which binds both chaperones. After transferring the steroid receptor to Hsp90, Hsp70 dissociates and is replaced by co-chaperones such as p23 and the high molecular weight immunophilin FKBP52. It is only in this complex that the steroid receptor is activated to bind ligand with high affinity. |
And thirdly, which proteins does Hsp90 interact with. Again from The Jackson Laboratory:
|
Hsp 90 and Proteins it interacts with.
The abundant protein Hsp90 is thought to assist in the activation and assembly of specific proteins. Many of these proteins are critical for signal transduction and cell division. As a result, Hsp90 is a target for anti-tumour drugs.
|
|
 |
Other examples are: PDK1, PKC-gamma, vSRC/cSRC, PPAR-alpha and p53. A comprehensive list of interacting proteins compiled by Picard Laboratory can be found here.
Thus, looking at all the proteins that can be affected by Hsp90, there is no surprise that the protein is highly conserved and that effects are profound when Hsp90 is fiddled with.


